Meningococcal disease
is a rare, but potentially devastating bacterial infection2,3,4
- Can progress rapidly and be hard to diagnose5,6
- Early symptoms can be easily mistaken for a common cold or flu6
- Most children survive but if not diagnosed and treated quickly it can lead to death within 24 hours 2,3,5,6,
Meningococcal B, W and Y strains cause the majority of disease in Australia, however…7
MenBMeningococcal B is currently the most
predominant strain in Australia7

Australia, 1997-2018, by year and bacterial serogroup
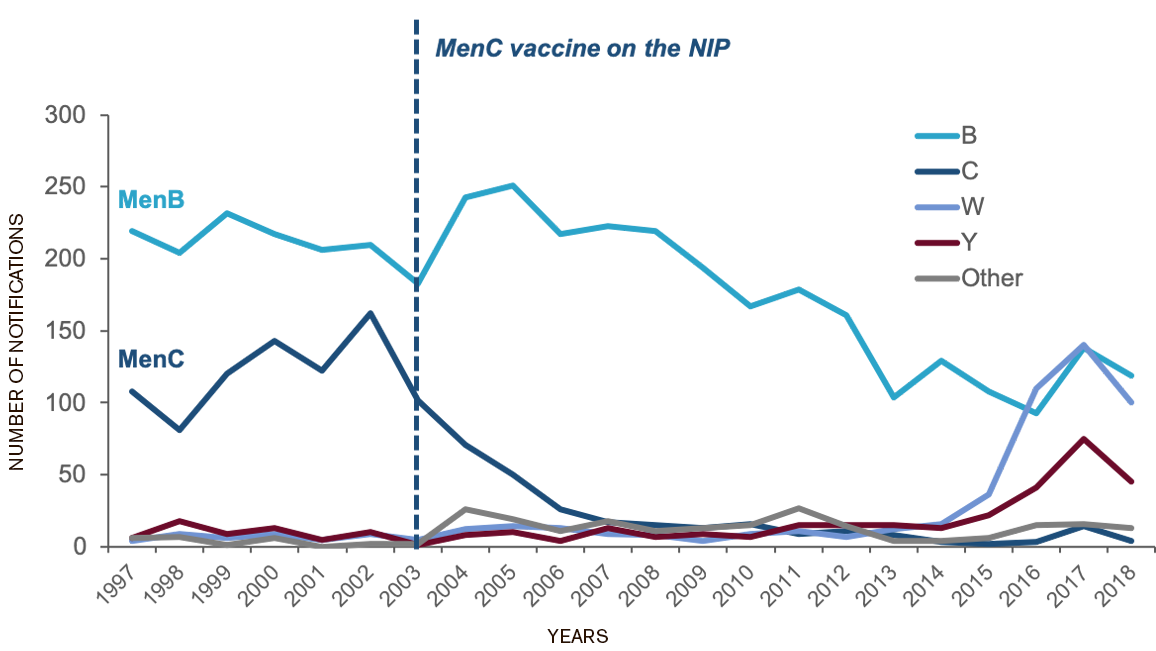
Adapted from Annual reports of the meningococcal surveillance program9 and the Department of Health, 201810
Note: 'Other' includes other serogroups not signed out, cases where meningococcal isolates could not be identified, other isolates not grouped and cases where serogroup was not known.
MenBMeningococcal B is the most common strain
in infants and adolescents9

Infants are most at risk of MenB, followed by adolescents8
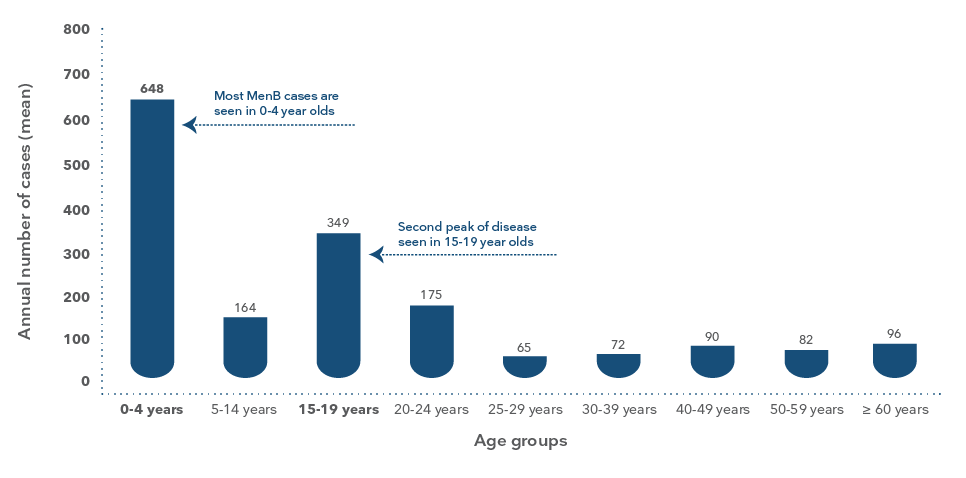
Age specific total numbers of invasive meningococcal B disease in all age groups, Australia 2006-20158
Adapted from Archer BN et al. Med J Aust 2017; 207(9): 382-7
MenBMeningococcal B has a peak incidence of
disease in those aged 3-6 months8

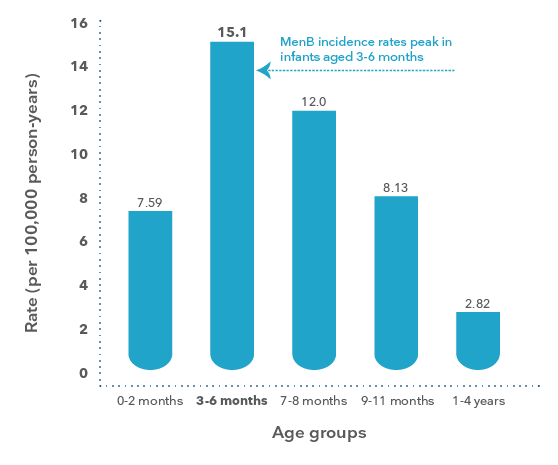
Age specific (0-4 years) rates of invasive meningococcal B disease in Australia 2006-20158
Adapted from Archer BN et al. Med J Aust 2017; 207(9): 382-7
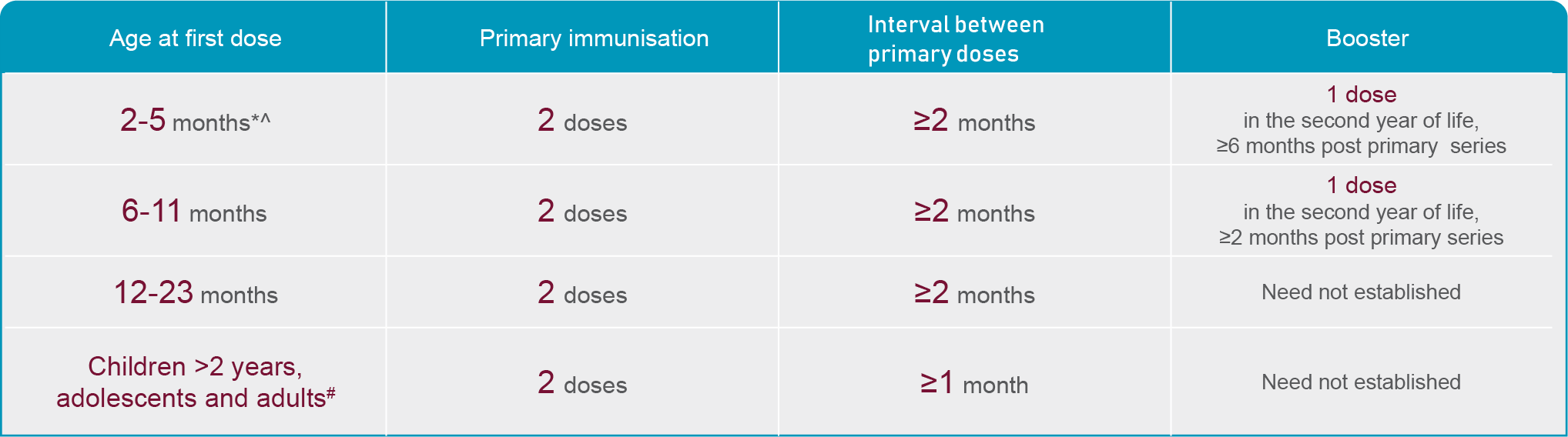
Meningococcal B vaccination is strongly recommended in the Australian Immunisation Handbook for the following groups:3


*Laboratory personnel that frequently handle N. Meningitidis, any person with certain medical conditions that increase their risk of IMD. See handbook for more details.
Note: Bexsero is indicated from 2 months of age1
Parents want to know about MenB11
IPSOS market research suggests that “9 out of 10 Australian parents expect their family doctor to initiate discussions around all strains of meningococcal disease.” Hear from Jenna's family…
Expert advice from Dr Lisa Beecham
How to talk to parents about meningococcal disease and vaccination
“Most people can find the money if they know that it is an important vaccination…”
Hear tips from Dr Lisa Beecham on proactively initiating discussion on Meningococcal B vaccination among parents of infants
Information on Bexsero (MenB Vaccine)
- BEXSERO Approved PRODUCT INFORMATION.
- Rosenstein NE, et al. N Engl J Med. 2001; 344:1378-1388
- Australian Technical Advisory Group on Immunisation (ATAGI). Australian Immunisation Handbook, Australian Government Department of Health, Canberra, 2018, immunisationhandbook.health.gov.au.
- Viner RM, et al. Lancet Neurol. 2012; 11:774-783
- van Deuren M, et al. Clin Microbiol Rev 2000; 13(1):144-66
- Thompson MJ et al. Lancet 2006; 367(9508):397-403
- Department of Health. INVASIVE MENINGOCOCCAL DISEASE NATIONAL SURVEILLANCE REPORT 1 January to 30 June 2019. https://www1.health.gov.au/internet/main/publishing.nsf/Content/5FEABC4B495BDEC1CA25807D001327FA/$File/IMD-surveil-report-Jan-June-2019.pdf
- Archer BN, et al. Med J Aust 2017; 207(9):382-7
- Annual reports of the Australian Meningococcal Surveillance programme, 1997-2018 http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-pubs-annlrpt-menganrep.htm [Accessed January 2018]
- Department of Health. Invasive Meningococcal Disease National Surveillance Report. Quarter 4, 2018. 1 October to 31 December 2018 http://www.health.gov.au/internet/main/publishing.nsf/Content/5FEABC4B495BDEC1CA25807D001327FA/$File/1Oct-31Dec19-qrt3-IMD.pdf [Accessed April 2019]
- Data on file IPSOS. National Meningococcal Disease Awareness Campaign Survey, conducted on behalf of GSK. Australia, September 2018.
- BEXSERO Summary of Product characteristics. GSK Vaccines S.r.l.;2018
- Martinón-Torres F, Safadi MAP, Martinez AC, et al. Reduced schedules of 4CMenB vaccine in infants and catch-up series in children: immunogenicity and safety results from a randomised open-label phase 3b trial. Vaccine. 2015: 33(32):3850-3858
- Vesikari T, Prymula R, Merrall E, Kohl l, Toneatto D, Dull PM, Meningococcal serogroup B vaccine (4CmenB): booster dose in previously vaccinated infants and primary vaccination in toddlers and two-year old children. Vaccine. 2015: 33(32): 3850-3858
- Watson PS, Turner DPJ. Clinical experience with the meningococcal B vaccine, Bexsero prospects for reducing the burden of meningococcal serogroup B disease. 2016: 34(7): 875-880
- Martinón-Torres F et al. J Infect 2018; 76:258-269
- EMA 2018. CHMP meeting minutes, April 2018. https://www.ema.europa.eu/en/documents/agenda/agenda-chmp-agenda-23-26-april-2018-meeting_en.pdf
- Pfizer Ltd. Trumenba. Annex I: Summary of product characteristics. EMA: May 2018
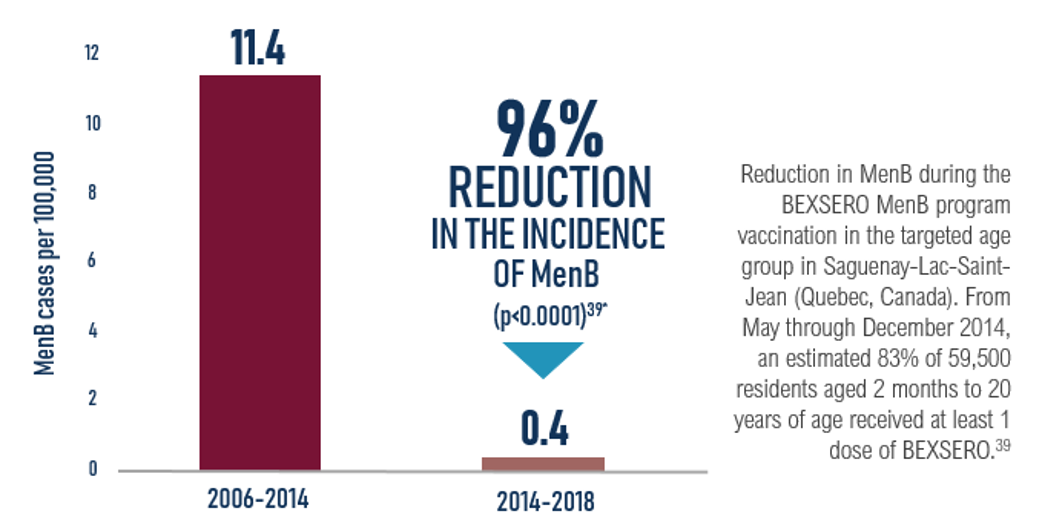
- Epidemiological impact of the vaccination campaign against serogroup b meningococcal disease in the saguenay-lac-saint-jean region in 2014: report of 30 june 2018
- Watson PS, Turner DPJ. Clinical experience with the meningococcal B vaccine, Bexsero®: prospects for reducing the burden of meningococcal serogroup B disease. Vaccine. 2016;34(7):875-880.
- GSK. Data on File – 2016N297580_02
- Meningococcal B. Health and Safety Executive (HSE). National Immunization office. Dublin, Ireland.
- Piano Nazionale Prevenzione Vaccinale PNPV 2017-2019. Ministero della salute website.
- Moreno-Pérez D, Álvarez Garcia FJ, Aristegui Fernández J, et al: for the CAV-AEP. Calendario de vacunaciones de la Asociación Española de Pediatria (CAV-AEP): recomendaciones 2016. Asociacion Española de Pediatria. [Immunisation schedule of the Spanish association of Paediatrics: 2016 recommendations]. An Pediatr (Barc). 2016 Jan; 84(1): 60. E1-13.
- Australian Technical Advisory Group on Immunisation (ATAGI) Statement.
- Impfplan Österreich 2018. Bundesministerium für Gesundheit website.
- Sociedade Brasileira de Imunizações. Informe Técnico 25/05/2015-Vacina meningocócica B.
- Advice for the use of the multicomponent meningococcal serogroup B (4CMenB) vaccine. An Advisory Committee Statement (ACS). National Advisory Committee on Immunization (NACI). April 2014. Public Health Agency of Canada website.
- Doporucení Ceské vakcinologické spolecnosti pro ockování proti invazivním meningokokovým onemocnením. Ceská vakcinologická spolecnost CLS JEP website.
- Clinical recommendation of the Pediatric Society-Greece.
- Melles M. Epidemiológiai információs hetilap. Országos Epidemiológiai Központ; 2014;22:1-60.
- Programu Szczepień Ochronnych na rok 2018. Dziennik Urzçdowy Ministra Zdrowia website.
- Recomendações sobre vacinas extra programa nacional de vacinação: Atualização 2018.
- JCVI position statement on use of Bexsero® meningococcal B vaccine in the UK. March 2014.
- Lietuvos Respublikos Sveikatos Apsaugos Ministras. Isakymas Del Lietuvos Respublikos Svikatos Aspaugos Ministro 2015 M. Birzelio 12 D. Isakymo NR. V-757, “Del Lietuvos Respublikos Vaiku Profilaktiniu Skiepjimu Kalendoriaus Patvirtinimo” Pakeitimo. 2018. Vilnius, Lithuania.
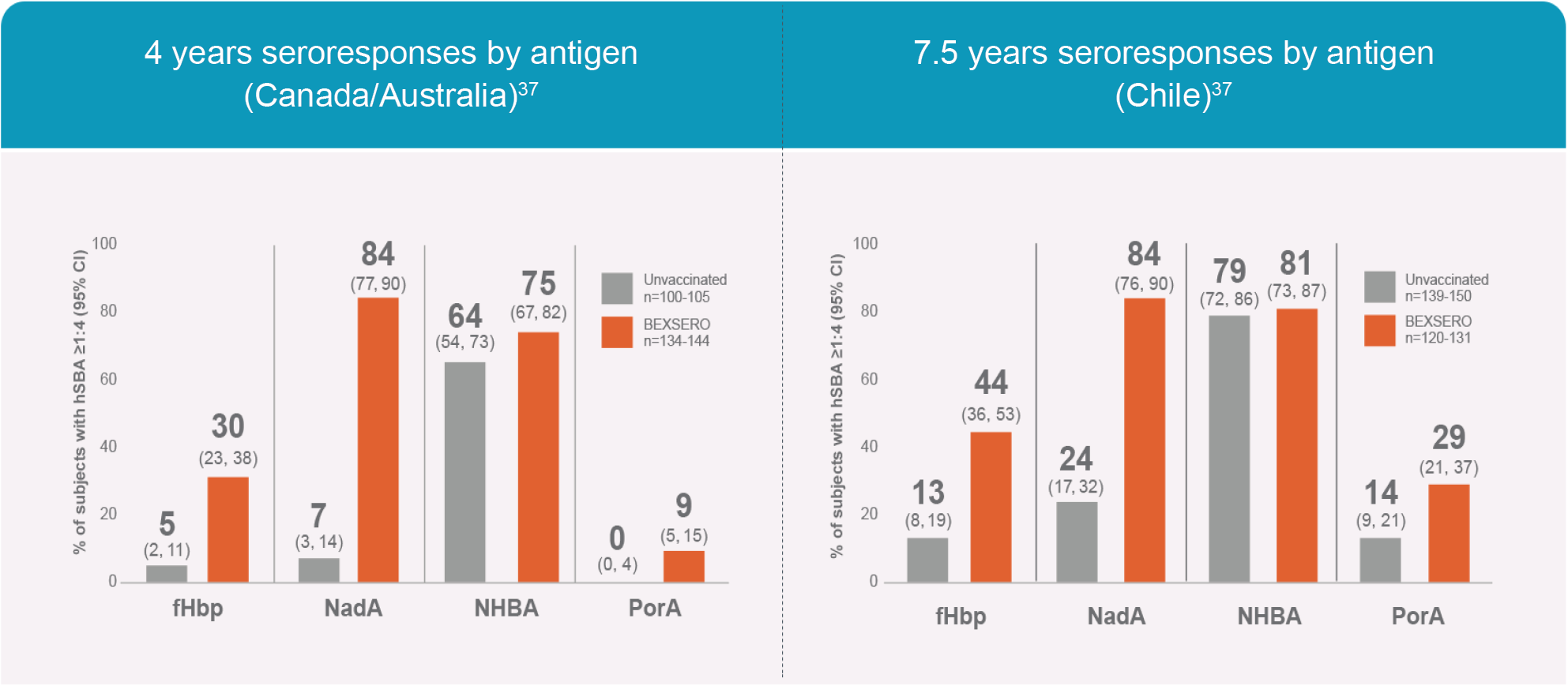
- Santolaya ME, O’Ryan ML, Valenzuela MT, et al. Immunogenicity and tolerability of a multicomponent meningococcal serogroup B (4CMenB) vaccine in healthy adolescents in Chile: a phase 2b/3 randomised, observer-blind, placebo-controlled study. Lancet. 2012;379(9816):617-624.
- Nolan T, Santolaya ME, de Looze F, et al. Antibody persistence and booster response in adolescents and young adults 4 and 7.5 years after immunization with 4CMenB vaccine. Vaccine. Available online 26 January 2019. https://doi.org/10.1016/j.vaccine.2018.12.059.
- Dretler AW, Rouphael NG, Stephens DS. Progress toward the global control of Neisseria meningitidis: 21st century vaccines, current guidelines, and challenges for future vaccine development. Hum Vaccin lmmunother. 2018;14(5):1146-1160.
- Deceuninck, G. et al. Vaccine 2019 https://doi.org/10.1016/j.vaccine.2019.06.021
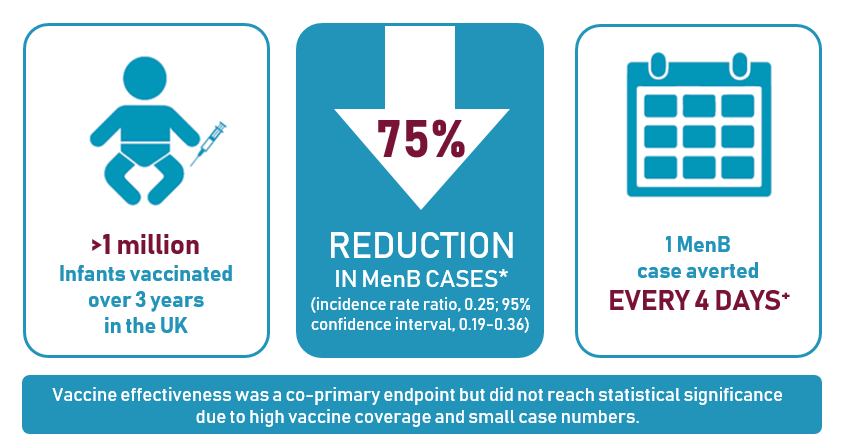
- Ladhani S et al. N Engl J Med 2020;382:309–317
Please review Bexsero Product Information before prescribing.
Full Product Information is available at au.gsk.com
For more information on this product please contact us.
Trade marks are owned by or licensed to the GSK group of companies.
© 2020 GSK group of companies or its licensor.
PM-AU-BEX-WCNT-200005 Date of GSK Approval: September 2020