WHY INVESTIGATE DTG/RPV AS A 2-DRUG REGIMEN?

STUDY DESIGN
INCLUSION CRITERIA

PATIENT DEMOGRAPHICS

PRIMARY ENDPOINT RESULT

BARRIER TO RESISTANCE

VIROLOGIC WITHDRAWALS

ADVERSE EVENTS

BONE MARKERS

LIPIDS

References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
Why investigate DTG/RPV as a 2-drug regimen?
- The requirement for life-long ART for HIV infection has highlighted a need to minimise cumulative drug exposure[5-7]
- The potency, safety, and resistance barrier of DTG makes it an ideal core agent for 2DR[3], [8]
- The safety, tolerability, and efficacy of RPV make it an optimal partner[9]
- The SWORD-1 & -2 studies evaluated whether a 2DR of DTG + RPV once daily was as effective as a traditional 3 or 4 drug regimen for the maintenance of virologic suppression[1]
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- The Antiretroviral Therapy Cohort Collaboration. Lancet HIV 2017;4:e349-56.
- Marcus JL, et al. JAIDS 2016;73:39-46.
- Raffi et al. HIV Med 2016;17(suppl 5):3-16.
- Ford et al. Antimicrob Agents Chemother 2013;57:5472-5477.
- Palella et al. AIDS 2014;28:335-344.
Study Design[1], [5]
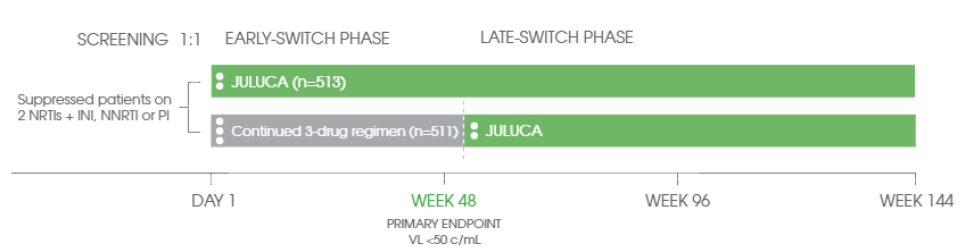
*8% non-inferiority margin for pooled data. 10% non-inferiority margin for individual studies
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Llibre et al. CROI 2017; Seattle, WA. Abstract 2421.
Inclusion Criteria[1], [5],
*2NRTIs + NNRTI; 2NRTIs + INI; 2 NRTIs + PI
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Llibre et al. CROI 2017; Seattle, WA. Abstract 2421.
Patient Demographics[1], [5]
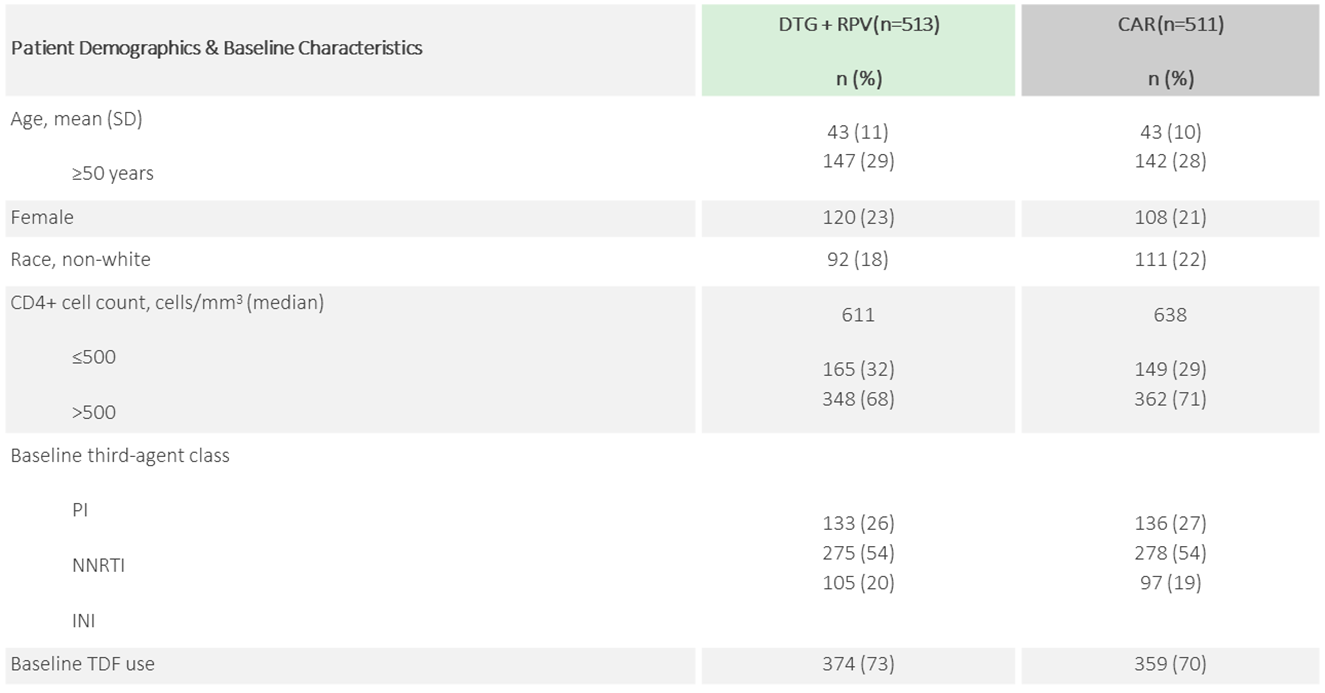
Data pooled across SWORD-1 and -2
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Llibre et al. CROI 2017; Seattle, WA. Abstract 2421.
Primary Endpoint Result
JULUCA—95% of Patients Maintained Virological Suppression[1]
Snapshot virological outcomes at Week 48 (pooled)
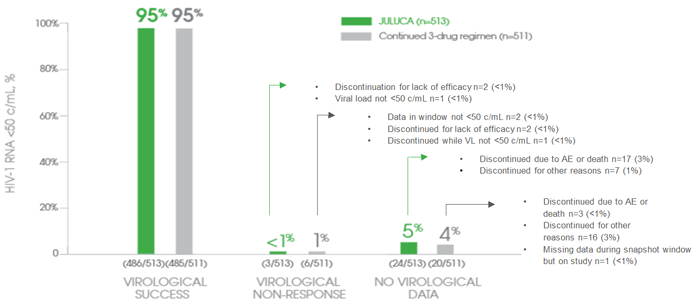
Non-inferiority was demonstrated in the pooled and individual analyses[1]

*Adjusted for age and baseline 3rd agent.
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
JULUCA—High Barrier to Resistance up to 48 Weeks
For patients switched to JULUCA, there was no increased risk of virological failure vs continued 3-drug regimen (<1% vs 1%, respectively)[1]

- One NNRTI mutation was identified (K101K/E) in a subject with documented poor/non-adherence; however, the virus remained susceptible to RPV[1]
One Subject with an Identified NNRTI Mutation After Poor/Non-Adherence Resuppressed on DTG + RPV at Week 45
- 41-year-old female participant randomised to DTG + RPV[5]
- Viral load history: pre-treatment >2 million copies/mL; at EVF/TDF/FTC initiation 968,000 copies/mL[5]
- Documented poor/non-adherence leading up to Week 36[5]

References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Llibre et al. CROI 2017; Seattle, WA. Abstract 2421.
Virologic Withdrawals[1], [5],

- One subject on DTG + RPV meeting virologic withdrawal criteria had identified an NNRTI resistance–associated mutation (K101K/E)
- No INI resistance–associated mutations were identified
*Data pooled across SWORD-1 and SWORD-2.
**Confirmed virologic withdrawals – Current “retest” HIV-1 RNA ≥200 c/mL, prior ≥50 c/mL.
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Llibre et al. CROI 2017; Seattle, WA. Abstract 2421.
Adverse Events
No new safety signals were identified for DTG + RPV
- Patients switched to JULUCA had a median time on ART of 4.25 years[1]
- 87% of subjects were new to both DTG and RPV[5]
Adverse events (AEs) with onset through Week 52 (pooled)[6]

To learn more in relation to the safety and tolerability of JULUCA click here
*Two deaths in the study, both unrelated to study drug. DTG + RPV: Kaposi's sarcoma (n=1), continued 3-drug regimen: lung cancer (n=1).
Reference:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Oglesby A, Punekar Y, Angelis K, et al. EACS 2017; Milan, Italy. Poster BPD1/2.
- Llibre et al. CROI 2017; Seattle, WA. Abstract 2421.
Bone Markers
JULUCA—Significant Recovery in Bone Mineral Density (Dexa Sub-Study)[5]
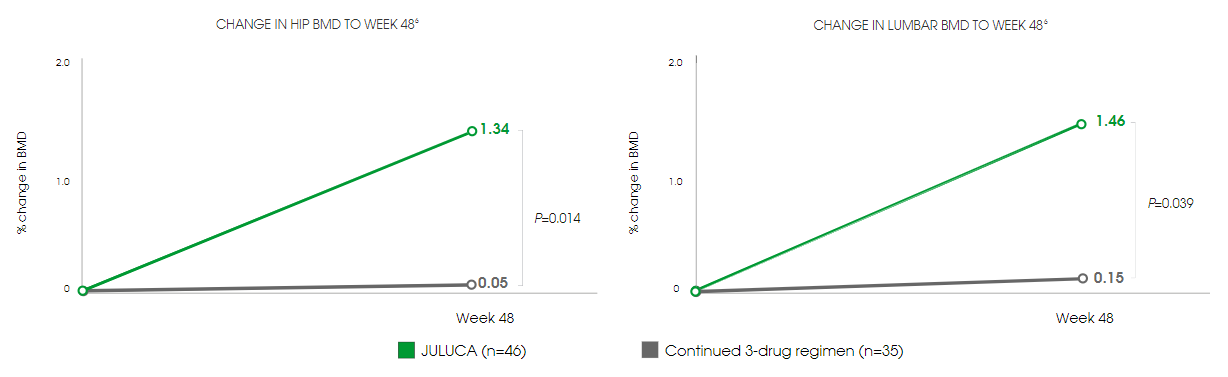
- 1.29% and 1.32% improvement in hip and lumbar spine bone mineral density (BMD), respectively, compared with those continuing on TDF-based regimens[5]
- Significant decrease in all measured markers of bone turnover, compared with those continuing on TDF-based regimens[5]
Switching to JULUCA provides a robust option for maintaining virological suppression while preserving bone health.
Bone Health & HIV
Effect of DTG + RPV on Bone Mineral Density in SWORD Studies
Reference:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- McComsey G, Gonzalez-Garcia J, Lupo S, et al. Sub-study 202094 of SWORD-1 and SWORD-2: switch from TDF-containing regimen to DTG + RPV improves bone mineral density and bone turnover markers over 48 weeks. Presented at: Annual International AIDS Conference; July 23-26, 2017; Paris, France. Poster TUPDB0205LB.
JULUCA—Maintains Lipid Levels
Despite the majority of patients switching away from TDF-based regimens, lipid values remained stable at Week 48[1]

No clinically relevant difference in renal parameters in either treatment arm[1]
- Improvements in some renal biomarkers, suggesting a favourable effect on renal tubular function
- No change in eGFR in either group as measured by cystatin C
References:
- Llibre JM, et al. Lancet 2018;391:839–849.
- McComsey GA, et al. AIDS 2018;32:477–485.
- Tivicay▼ (dolutegravir) Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.
- Rilpivirine Summary of Product Characteristics. Available from: www.medicines.ie. Last accessed: January 2019.



 Download the SWORD Slide Set
Download the SWORD Slide Set Access the SWORD Clinical Paper
Access the SWORD Clinical Paper