Meet the patients you can protect1, 5-7
Nearly everyone ≥50 years old needs shingles protection
Not a healthcare professional? Visit our Public site
Not a healthcare professional? Visit our Public site

59
The same results were first published in the New England Journal of Medicine.*
*The graph has been independently created by GSK from the original data.

Pooled data from ZOE-50 (subjects ≥50 years old) and ZOE-70 (subjects ≥70 years old). PHN was defined as HZ-associated pain rated as ≥3 on a 0-10 scale, occurring or persisting for more than 90 days following the onset of rash using the Zoster Brief Pain Inventory questionnaire.1

REDUCTION OF THESE SHINGLES-RELATED COMPLICATIONS2
The evaluated HZ-related complications were: HZ vasculitis, disseminated disease, ophthalmic disease, neurologic disease, visceral disease, and stroke. In the pooled analysis of ZOE-50 and ZOE-70, SHINGRIX▼ significantly reduced these HZ-related complications by 93.7% (95% CI: 59.5; 99.9) and 91.6% (95% CI: 43.3; 99.8) in adults ≥50 years (1 vs.16 cases) and adults ≥70 years (1 vs.12 cases), respectively.
Study Design: Pooled data from the ZOE-50 phase 3 trial (subjects ≥50 years old) and the ZOE-70 phase 3 trial (subjects ≥70 years old)—2 placebo-controlled, observer-blind, phase 3 trials conducted in 18 countries among immunocompetent individuals who were randomized to receive 2 doses (0 and 2 months) of either SHINGRIX▼ or placebo. The primary efficacy analysis included all subjects who did not develop a confirmed case of herpes zoster within 1 month after the second dose.1-4
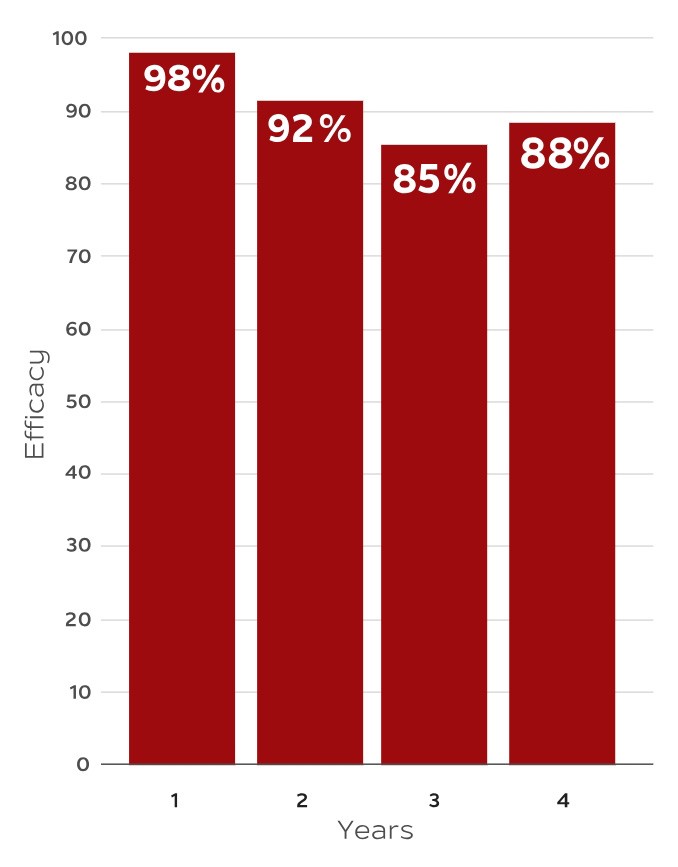
*Cases/1000 person-years in vaccinated versus placebo are 0.2 vs 10.1, 0.9 vs 11.1, 1.2 vs 7.7, and 1.0 vs 8.2 for year 1, 2, 3, and 4, respectively (P<0.001).1
†Number of patients vaccinated vs received placebo are 8250 vs 8346, 8039 vs 8024, 7736 vs 7661, 7426 vs 7267 for year 1, 2, 3, and 4, respectively.1
Data shown in the graph represents subjects ≥70 years of age from the pooled analysis of ZOE-50 and ZOE-70. The efficacy of SHINGRIX▼ continues to be monitored.1,2
The same results were first published in the New England Journal of Medicine.‡
‡The graph has been independently created by GSK from the original data.
CI=confidence interval; HZ=herpes zoster; PHN=postherpetic neuralgia.
Shingrix▼ Safety Information2
Contraindications:
Hypersensitivity to the active substances or to any component of the vaccine.
Special warnings and precautions:
As with all injectable vaccines:
- appropriate medical treatment and supervision should always be readily available in case of an anaphylactic event following the administration of the vaccine;
- vaccination with Shingrix▼ should be postponed in subjects suffering from an acute severe febrile illness. The presence of a minor infection, such as a cold, should not result in the deferral of vaccination;
- a protective immune response may not be elicited in all vaccinees.
Do not administer the vaccine intravascularly, intradermally or subcutaneously.
There are no data on the use of Shingrix▼ in pregnant women and the effect on breast-fed infants of administration of Shingrix▼ to their mothers has not been studied.
Adverse reactions:
- Very common (≥1/10): headache, gastrointestinal symptoms (including nausea, vomiting, diarrhoea and/or abdominal pain), myalgia, injection site reactions (such as pain, redness, swelling), fatigue, chills, fever.
- Common (≥1/100 to <1/10): injection site pruritus, malaise.
Please refer to the full prescribing information for further details.
References
▼This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. To report Adverse Event/s associated with the use of GSK product/s, please contact us via saudi.safety@gsk.com
For more information, please refer to the prescribing information or contact GSK: P.O Box 55850, Jeddah, 21544, Kingdom of Saudi Arabia.
Telephone: +966 12 653 6666 or via gcc.medinfo@gsk.com
To report Adverse Event/s associated with the use of GSK product/s, please contact us via saudi.safety@gsk.com
To report the Quality related product complaint/s associated with the use of GSK product/s, please contact us via ksa.productqualitycomplaint@gsk.com
GSK does not recommend, endorse or accept liability for sites controlled by a third party.
Trademarks are owned by or licensed to the GSK group of companies.
©2025 GSK group of companies or its licensor
GlaxoSmithKline, Jeddah, 21544, Kingdom of Saudi Arabia.
PM-SA-SGX-WCNT-240011 Date of preparation: September 2025